| Crystal system | Orthorhombic |
| Transparency | Transparent to opaque |
| Luster | Vitreous |
| Fracture | Uneven to sub-conchoidal |
| Cleavage | Perfect prismatic |
| Specific Gravity | 3.45 |
| Hardness | 4.5-5 |
| Optical Character | Biaxial + ; Double Refractive |
| Refractive index | 1.614-1.636 |
| Birefringence | 0.022 |
| Dispersion | |
| Fluorescence | |
| Pleochroism | |
| Chemical Formula | Zn4(OH)Si2O7.H2O |
| Comments | Soluble in HCl but does not effervesce |
| Streak | White |
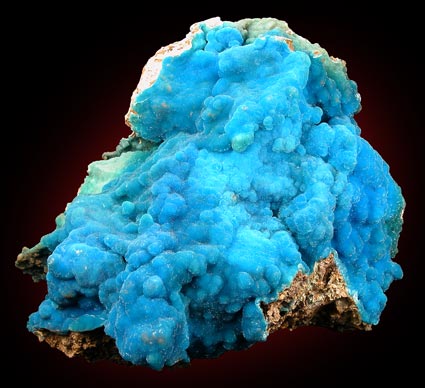
Although hemimorphite (hem-ah-MORE-fite) is an important ore of zinc (a zinc silicate, corresponding to the formula Zn4(OH)Si2O7.H2O), it is only rarely fashioned for gem use. The orthorhombic crystals are usually thin and tabular or pyramidal in habit, sometimes showing an obvious hemimorphic development; i.e., unsymmetrical as regards the two ends of the crystals. This, in fact, is the origin of the mineral's name. It is usually colorless but may be green, blue or brown, sometimes banded. More commonly, it occurs in fibrous, globular, granular or porous and earthy masses; sheaf like or crested groups are also found.
Hemimorphite has two directions of perfect prismatic cleavage, a hardness of 4 1/2 to 5, an S.G. of 3.40 to 3.50, a vitreous luster, an uneven to sub-conchoidal fracture, a white streak, and varies from transparent for the crystals to opaque for the massive material. It fuses with difficulty but gelatinizes easily with acids. The R.I. is about 1.614 - 1.636; the double refraction is biaxial in character and positive in sign. It may be distinguished from smithsonite, which it resembles, by the effervescence of smithsonite with hydrochloric acid.
This mineral is a secondary product, formed by the action of silica bearing water on other zinc ores. It is usually found in limestones associated with smithsonite, sphalerite, galena, cerussite and anglesite. Often it is intimately intermixed with smithsonite. A few of the many localities of hemimorphite are Aachen, Germany, Raibel and Bleiberg, Carinthia; Austria; Silesia; Cumberland and Derbyshire, England; Sardinia; Mexico; Sussex County, New Jersey; and Phoenixville and Friedensville, Pennsylvania.